|
Evolution of Parasite Resistance
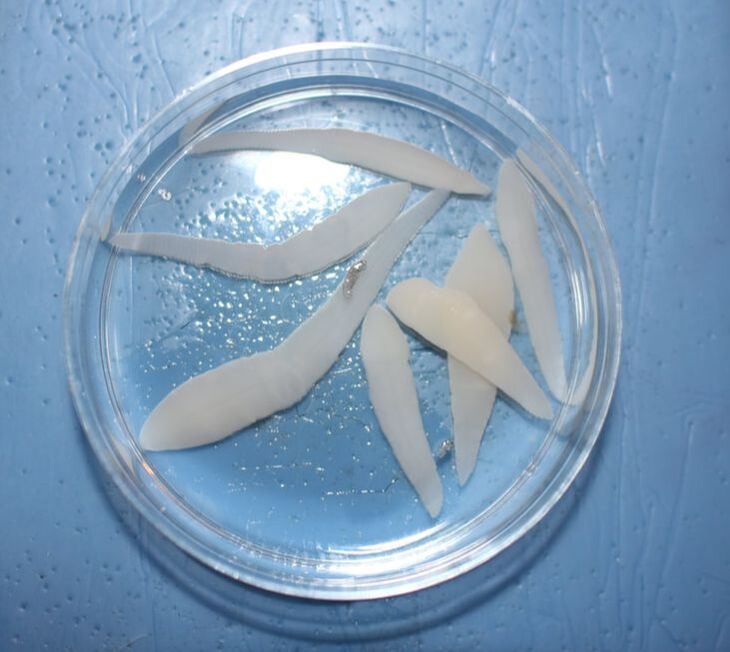
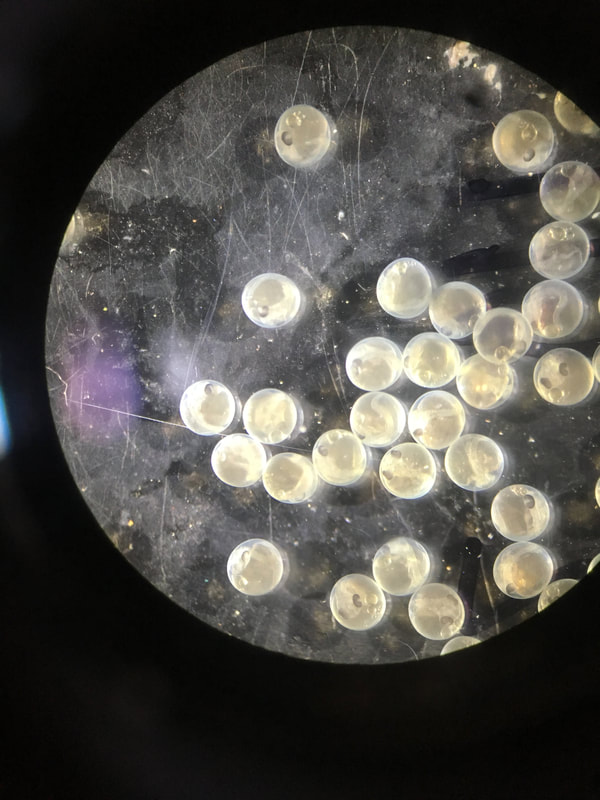
Infection outcomes are determined by a complex set of interactions between host immunity and parasite infectivity. These host and parasite traits often have a genetic basis and can coevolve rapidly, leading to large variation in infection outcomes between individuals and across populations. In work I started as a postdoc Dr. Dan Bolnick, I am exploring the details of host-parasite interactions using the threespine stickleback and it's tapeworm parasite, Schistocephalus solidus. We have identified fish populations that vary in their parasite resistance, which is largely determined by the ability of fish to produce fibrosis tissue when infected that surrounds and inhibits the tapeworm. Using field samples and gene expression data from both hosts and tapeworms, I am working to understand how host and parasite strategies evolve, how multiple tapeworms interact within the same host, and to identify genes that interact across species boundaries. |